Interesting facts from the world of organic chemistry
Derivatives - substituted derivatives of carboxylic acids:
PABA
4-aminobenzoic acid, formerly p-aminobenzoic acid (hence the abbreviation PABA), is a relatively simple compound but with significant properties. Due to its ability to willingly absorb short-wave UV radiation and thereby help protect the skin from the negative effects of the sun’s rays, PABA was used in sunscreens in the past. Only later was it discovered that PABA increases the formation of DNA defects in skin cells and thereby increases the risk of skin cancer. That is why we only rarely encounter it in sunscreens.
PABA is an important (essential) nutrient for some bacteria. PABA is needed for the production of folic acid, which is further needed to maintain the growth of healthy cell walls (bacterial vitamin H1). For the human body, PABA is a biologically active substance sometimes classified as a vitamin (Bx). Officially, it is not one of the vitamins, because its production in the body is ensured by the bacteria Escherichia coli living and thriving in the large intestine. The production of this vitamin is also mediated by the bacterium Lactobacillus lactis according to the following scheme.
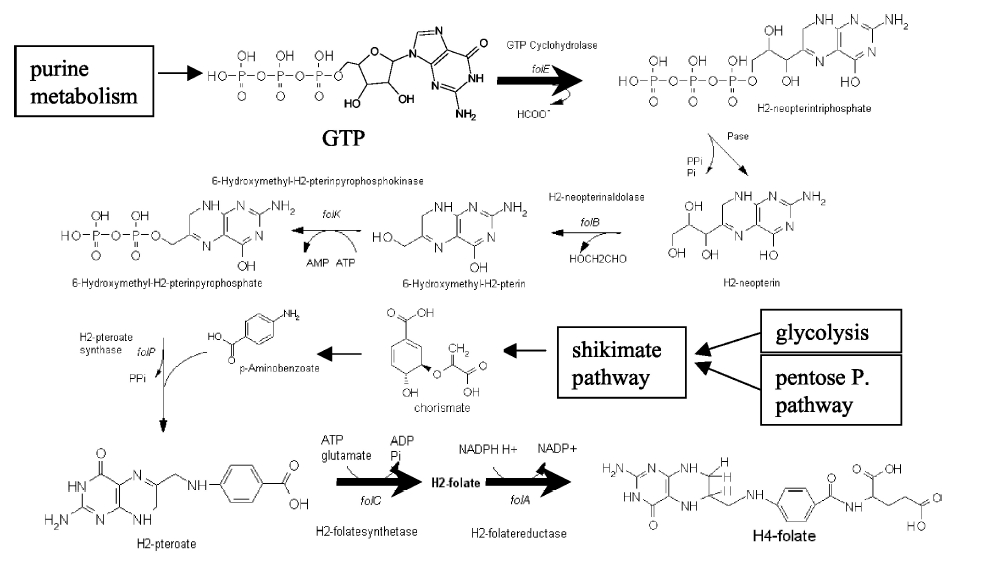
Picture above: Biosynthesis of 4-aminobenzoic acid (PABA) by Lactobacillus lactis bacteria.
In laboratory conditions, PABA can be prepared from toluene after its nitration and separation of 2-nitrotoluene. This gives us 4-nitrotoluene, which is converted to 4-methylaniline by reacting with Fe and HCl. In the next step, we perform a reaction with acetic anhydride, oxidation with potassium permanganate, and after acid hydrolysis we have the desired product. The reaction sequence is shown in figure below.
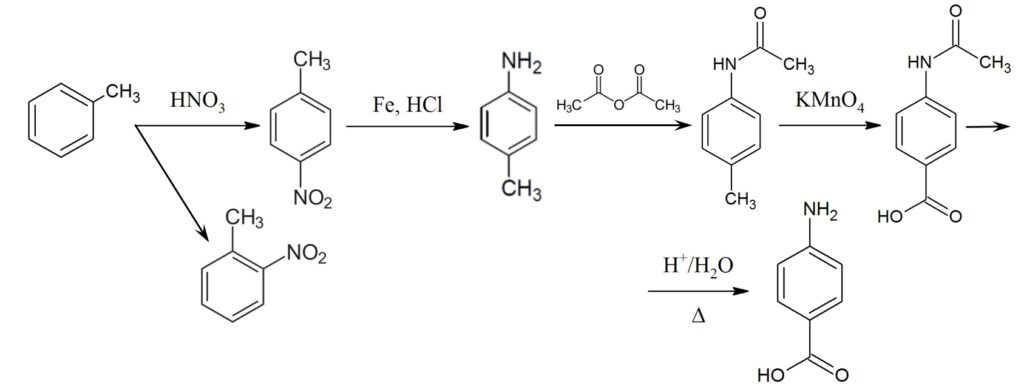
Picture above: Synthesis of PABA molecule in laboratory. The starting substance is toluene.
Sources:
[1] P. J. Osgood, S. H. Moss a D. J. Davies, „The sensitization of near-ultraviolet radiation killing of mammalian cells by the sunscreen agent para-aminobenzoic acid.,“ Journal of Investigative Dermatology, pp. 354-357, 1982.
[2] M. Doležal, „doc. Dr. Ing. Marek Doležal,“ 14 3 2013. [Online]. Available: http://web.vscht.cz/dolezala/CHPC/11%20Vitaminy_cvi%C4%8Den%C3%AD.pdf. [Přístup získán 8 6 2013].
[3] iGEM 2008, „California Institute of Technology,“ 2008. [Online]. Available: http://2008.igem.org/Team:Caltech/Project/Vitamins. [Přístup získán 8 6 2013].
